Research & Technology
Research and Technology
TargTex’s lead program on GBM is based on a machine learning algorithm that deciphered a relationship between an unexplored target for GBM – calcium channel – and a small molecule with known anticancer properties.
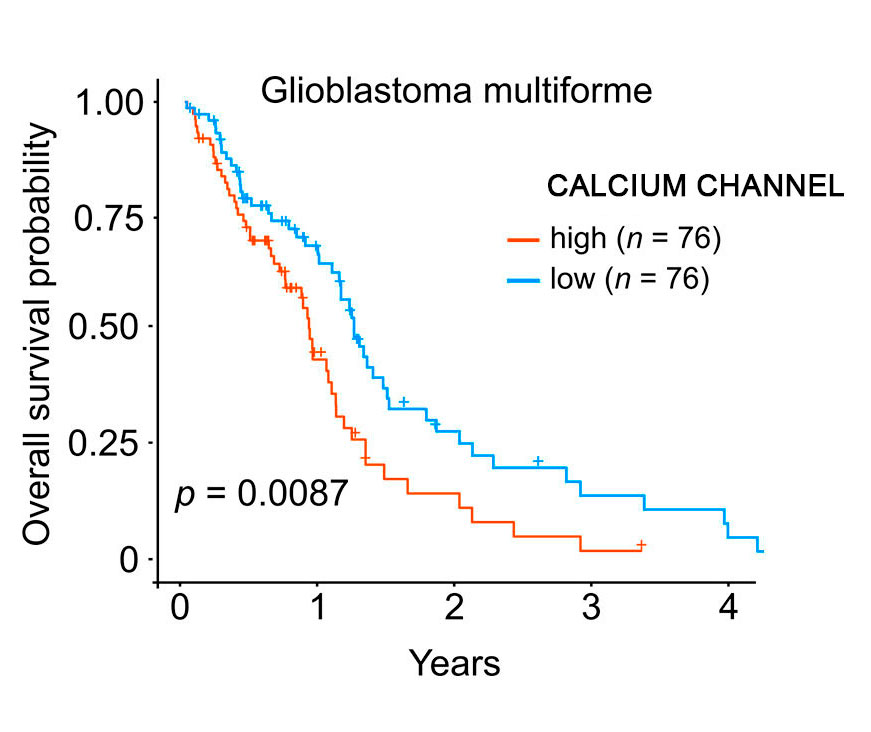
Calcium Channel overexpression in GBM
High calcium channel expression is strongly associated with poor survival in patients with gliomas, especially for Glioblastoma, which was confirmed by evaluating clinical data for 9,785 tumor samples belonging to 30 cohorts from The Cancer Genome Atlas.
Hydrogel Design for Local Administration
Given the chemical properties of the small molecule and the particularities of the pathology we engineered a hydrogel to be applied as an adjunct to surgery after tumor removal. This hydrogel was formulated to have the ideal pharmacokinetics to eliminate the remaining GBM cells after tumor removal and, thus, avoid recurrence.
Proof-of-concept
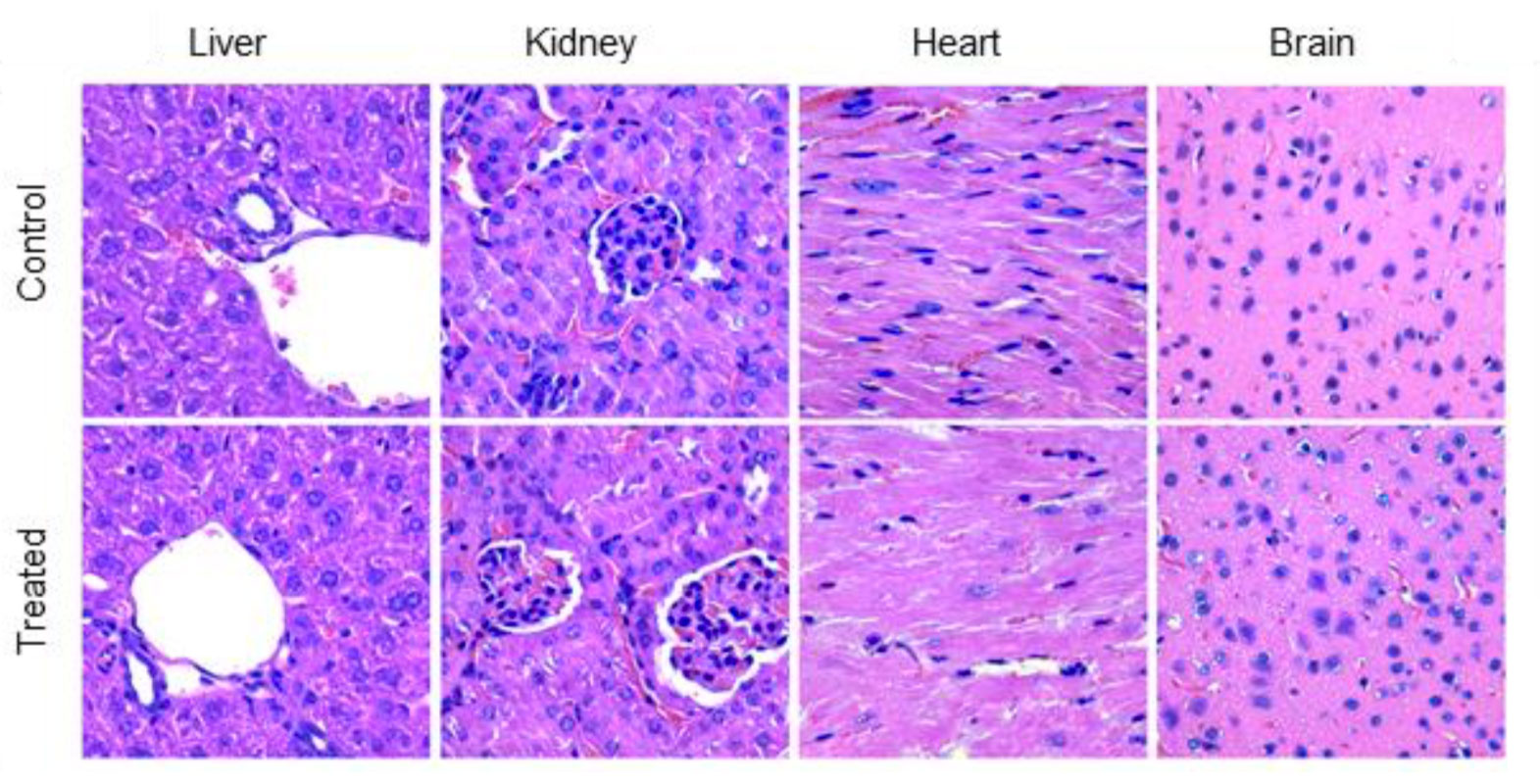
Our biocompatible hydrogel formulation was tested in mice where GBM cells were injected intracranially. The treated group successfully reduces tumor volume completely. Histopathology of liver, kidney, lung, spleen, gastrointestinal tract and heart showed no significant changes/evidence of toxicity. No evidence for necrosis or other lesions (e.g. inflammation) were seen in the brain or at the site of administration.
HealthCare Professionals
Considering the two major hurdles that we have identified to tackle GBM, which are common inability to achieve complete resection along with the blood brain barrier (BBB) crossing of therapeutic agents, TargTex has developed an adjunct to surgery product to address both issues.
Adjunct to surgery
TargTex is aware that other adjunct to surgery agents have shown issues of their own, such as added risk of infection and inflammation, impairment on tumor recurrence evaluation and difficult-to-handle solutions that leads to low adoption rates for this localized treatment.
Nevertheless, we are focused on developing our formulation alongside with experienced neurosurgeons to address these issues and to circumvent the necessity of crossing BBB. Due to its selectivity, TargTex’s product is sought to have less side effects and to act during the no-treatment hiatus between surgery and the standard-of-care treatment (radio- and chemotherapy).
Recurrent Surgery
Moreover, another positive feature of TargTex’s molecule is that it acts on a target that is even more overexpressed in recurrent GBM patients than in newly diagnosed GBM patients.
Therefore, and considering that clinical reports demonstrate a clear benefit of a second surgery over a non-surgical approach and that most GBM recurrence are within 2 cm of the original tumor site (70-90%), we envision an added value to our product in this even more aggressive stage of the pathology.
Patients and Caregivers
As internal policy, TargTex chooses to value one of the key elements of treating GBM, which are the patients themselves. We want to develop our product in line with the patients’ needs as well as considering recurrent GBM patients growing reservations and fears when exposed to the possibility of going through the standard-of-care procedure all over again.
Selective GBM Drug
While survival is obviously a top need, we are aware that patients also express their worries mostly to retain brain functions, maintain basic physical tasks and reduce pain (especially headaches). Our approach was designed to mitigate these concerns being that TargTex’s molecule is not a standard chemotherapy agent. It is selective for GBM and therefore preliminary data shows that it spares the healthy brain tissues. In turn, this is expected to result in less side effects.
Single Administration with no Additional Procedures
Furthermore, being used as an adjunct to surgery, the localized treatment will have a faster and more effective response. This single administration is intended to eliminate the remaining GBM cells that could not be fully removed during surgery and thus avoid recurrence.
TargTex is developing a strong preclinical package to support our claims that will allow our product to reach clinical trials in a near future.
Glioblastoma
Glioblastoma
GBM is the most common and deadliest type of malignant primary brain tumor and affects nearly a quarter of a million patients globally with a global incidence of 3-4 new cases per 100 000 people.
Despite the efforts made over the last decades, GBM remains incurable with survival rates lower than 5% after 2 years.
Most of the newly GBM diagnosed cases go through brain tumor resection, although, unfortunately, only a small proportion of patients are eligible for a complete resection. In turn, this leads to residual GBM cells that can regrow and cause recurrence.
To avoid this scenario, radio- and chemotherapy regimens are used to treat the remaining tumor cells. Unfortunately, the presence of the protective blood-brain barrier (BBB) impairs drug delivery rendering chemotherapy ineffective.
As a result, GBM recurrence is nearly inevitable.
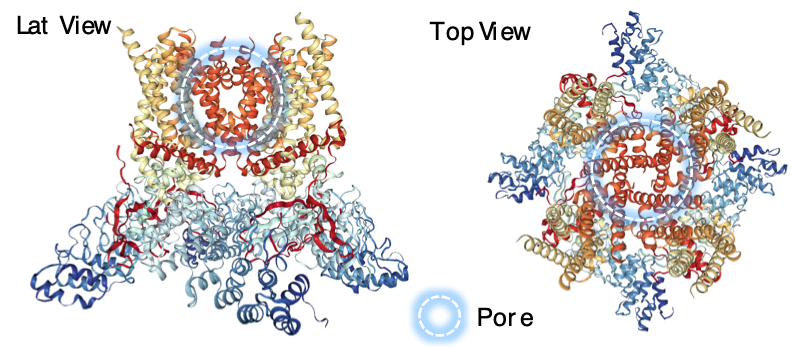
Calcium Channel
Calcium Channel
Calcium channel that is highly over-expressed in GBM tumor cells.
This cation channel is important for cellular calcium homeostasis and calcium (Ca2+) signalling. Calcium signalling plays an important role in cell death, proliferation and metastasis.
This calcium channel was found significantly higher in glioblastoma patients and its over-expression was related with a lower survival of the patients.
Solution
The vision
TargTex is focused on developing a targeted therapy for the treatment of Glioblastoma. Ultimately, TargTex will keep its focus on developing clinical candidates for different therapeutic indications, grounded on exploring new links between molecules of interest and relevant biological targets.
Strategy Planning
TargTex’s most advanced product is directed against Glioblastoma. We are fully dedicated on making this product the first successful clinical candidate that we plan to develop up to clinical trials phase II. Licensing agreements achieved after completing milestones that show clinical value of the product, will allow the company to re-invest in the pipeline and backup molecules that will be under development for other oncological indications.



